如果你也在 怎样代写统计力学Statistical mechanics这个学科遇到相关的难题,请随时右上角联系我们的24/7代写客服。
统计力学是一个数学框架,它将统计方法和概率理论应用于大型微观实体的集合。它不假设或假定任何自然法则,而是从这种集合体的行为来解释自然界的宏观行为。
statistics-lab™ 为您的留学生涯保驾护航 在代写统计力学Statistical mechanics方面已经树立了自己的口碑, 保证靠谱, 高质且原创的统计Statistics代写服务。我们的专家在代写统计力学Statistical mechanics代写方面经验极为丰富,各种代写统计力学Statistical mechanics相关的作业也就用不着说。
我们提供的统计力学Statistical mechanics及其相关学科的代写,服务范围广, 其中包括但不限于:
- Statistical Inference 统计推断
- Statistical Computing 统计计算
- Advanced Probability Theory 高等概率论
- Advanced Mathematical Statistics 高等数理统计学
- (Generalized) Linear Models 广义线性模型
- Statistical Machine Learning 统计机器学习
- Longitudinal Data Analysis 纵向数据分析
- Foundations of Data Science 数据科学基础
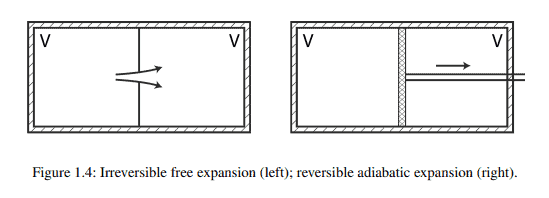
物理代写|统计力学代写Statistical mechanics代考|INTERNAL ENERGY: WORK AND HEAT
In the performance of adiabatic work $W_{\text {ad }}$-work on adiabatically isolated systems-it’s found that transitions $i \rightarrow f$ produced between reproducible equilibrium states ${ }^{8}(i, f)$ depend only on the amount of work and not on how it’s performed. Regardless of how the proportions of the types of work are varied, the same total amount of adiabatic work results in the same transition $i \rightarrow f$. This discovery is of fundamental importance. If the transition $i \rightarrow f$ produced by adiabatic work is independent of the means by which it’s brought about, it can depend only on the initial and final states $(i, f)$. That implies the existence of a physical quantity associated with equilibrium states that “couples” to adiabatic work, the internal energy $U$, such that ${ }^{9}$
$$
\Delta U=U_{f}-U_{i}=W_{\text {ad } .} .
$$
Internal energy is a state variable-one that depends only on the state of equilibrium of the system and not on how the system was prepared in that state. Adiabatic work done on a system increases its internal energy and is taken as a positive quantity. Adiabatic work done by a system (somewhere in the environment a weight is higher, a spring is compressed) is accompanied by a decrease in internal energy, and is taken as a negative quantity. Changes in internal energy come at the expense of adiabatic work done on or by a system. Equation (1.1) expresses conservation of energy: If we don’t let heat escape, work performed on the system is stored in its internal energy, energy that can be recovered by letting the system do adiabatic work on the environment. We’ll say that internal energy is the storehouse of adiabatic work.
Now, let work $W$ be performed under nonadiabatic conditions. It’s found, for the same transition $i \rightarrow f$ produced by $W_{\text {ad }}$, that $W \neq \Delta U$. The energy of mechanical work is not conserved in systems with diathermic boundaries. Energy conservation is one of the sacred principles of physics, and we don’t want to let go of it. The principle can be restored by recognizing different forms of energy. ${ }^{10}$ The heat transferred to or from the system, $Q$, is the difference in work
$$
Q \equiv \Delta U-W
$$
that effects the same change in state of systems with the two types of boundaries, $Q=W_{\text {ad }}-W$. If it takes more work $W$ to produce the same change of state as that under adiabatic conditions, $Q<0$ : heat leaves the system by flowing through the boundary; $Q>0$ corresponds to heat entering the system. ${ }^{11}$ Equation (1.2) is the first law of thermodynamics. One might think it applies to closed systems only, based on how we’ve formulated it. It applies to open systems when we introduce another kind of work-chemical work – the energy required to change the amount of matter in the system (see Section 1.6). The point here is that the nature of the boundaries allows us to classify different types of energy.
物理代写|统计力学代写Statistical mechanics代考| IRREVERSIBILITY, DISORGANIZATION
Entropy-discovered through an analysis of the second law of thermodynamics-is an unexpected yet significant development. ${ }^{17}$ The Clausius inequality is a consequence of the second law:[3, p33]
$$
\oint \frac{\mathrm{d} Q}{T} \leq 0,
$$
where the integral is over all steps of a cyclic process (one that returns a system to its initial state), $T$ is the absolute temperature 18 at which heat transfer $₫ Q$ occurs, and where equality in (1.6) holds for reversible heat transfers, ${ }^{19}$
$$
\oint \frac{(\mathrm{d} Q){\mathrm{rev}}}{T}=0 . $$ Differentials of state variables are exact. ${ }^{20}$ Exact differentials $\mathrm{d} g$ have the property that $\oint{C} \mathrm{~d} g=$ 0 for any integration path ${ }^{21} C$. We infer from Eq. (1.7) the existence of a state variable, entropy, the differential of which is ${ }^{22}$
$$
\mathrm{d} S \equiv(\mathbb{\pi} Q){\mathrm{rev}} / T . $$ The quantity $T^{-1}$ is the integrating factor ${ }^{23}$ for $(\mathrm{dQ}){\text {rev }}$. As a state variable, entropy is defined only in equilibrium. Changes in entropy between equilibrium states $(A, B)$ are found by integrating its differential, Eq. (1.8),
$$
S(B)-S(A)=\int_{A}^{B}(\mathrm{~d} Q)_{\mathrm{rev}} / T
$$ for any path connecting $A$ and $B$, such that heat transfers occur reversibly at all stages. ${ }^{24}$
统计力学代考
物理代写|统计力学代写Statistical mechanics代考|INTERNAL ENERGY: WORK AND HEAT
在绝热工作中的表现 $W_{\mathrm{ad}}$ – 研究绝热隔离系统 – 发现过渡 $i \rightarrow f$ 在可重现的平衡状态之间产生 ${ }^{8}(i, f)$ 仅取决于工 作量,而不取决于其执行方式。无论工种的比例如何变化,相同的绝热工作总量导致相同的转变 $i \rightarrow f$. 这一发 现具有根本性的重要性。如果过渡 $i \rightarrow f$ 绝热功的产生与产生它的方式无关,它只能取决于初始状态和最终状态 $(i, f)$. 这意味着存在与平衡状态相关的物理量,该物理量“耦合”到绝热功,内部能量 $U$ ,这样 ${ }^{9}$
$$
\Delta U=U_{f}-U_{i}=W_{\mathrm{ad} . .} .
$$
内能是一个状态变量,它仅取决于系统的平衡状态,而不取决于系统在该状态下的准备方式。对系统所做的绝热 功会增加其内部能量并被视为正数。系统所做的绝热功 (在环境中重量较高的地方,弹簧被压缩) 伴随着内部能 量的减少,并被视为负量。内部能量的变化是以系统上或系统完成的绝热工作为代价的。等式 (1.1) 表示能量守 恒: 如果我们不让热量逸出,系统所做的功将存储在其内部能量中,这些能量可以通过让系统对环境进行绝热工 作来回收。我们会说内能是绝热功的仓库。
现在,让工作 $W$ 在非绝热条件下进行。找到了,对于同一个过渡 $i \rightarrow f$ 由。。。生产 $W_{\mathrm{ad}}$ ,那 $W \neq \Delta U$. 在 具有透热边界的系统中,机械功的能量不守恒。能量守恒是物理学的神圣原则之一,我们不想放过它。这个原理 可以通过识别不同形式的能量来恢复。 ${ }^{10}$ 传入或传出系统的热量, $Q$ ,是工作的差异
$$
Q \equiv \Delta U-W
$$
这会影响具有两种类型边界的系统状态的相同变化, $Q=W_{\mathrm{ad}}-W$. 如果需要更多的工作 $W$ 产生与绝热条件 下相同的状态变化, $Q<0$ : 热量通过边界流出系统; $Q>0$ 对应于进入系统的热量。 ${ }^{11}$ 方程 (1.2) 是热力学 第一定律。有人可能会认为它仅适用于封闭系统,基于我们如何制定它。当我们引入另一种化学功一一改变系统 中物质数量所需的能量时,它适用于开放系统 (见第 $1.6$ 节) 。这里的重点是边界的性质使我们能够对不同类型 的能量进行分类。
物理代写|统计力学代写Statistical mechanics代考| IRREVERSIBILITY, DISORGANIZATION
熵一一通过对热力学第二定律的分析发现一一是一个出乎意料但意义重大的发展。 ${ }^{17}$ 克劳修斯不等式是第二定律 的结果: [3, p33]
$$
\oint \frac{\mathrm{d} Q}{T} \leq 0
$$
其中积分是循环过程的所有步骤(将系统返回到其初始状态的过程), $T$ 是传热的绝对温度 18 đ $Q$ 发生,并且 (1.6)中的等式适用于可逆热传递,19
$$
\oint \frac{(\mathrm{d} Q) \mathrm{rev}}{T}=0 .
$$
状态变量的微分是精确的。 ${ }^{20}$ 精确的差异 $\mathrm{d} g$ 拥有的财产 $\oint C \mathrm{~d} g=0$ 表示任何集成路径 ${ }^{21} C$. 我们从方程式推断。 (1.7) 状态变量樀的存在,其微分为 22
$$
\mathrm{d} S \equiv(\pi Q) \mathrm{rev} / T .
$$
数量 $T^{-1}$ 是积分因子 ${ }^{23}$ 为了 $(\mathrm{dQ}) \mathrm{rev}$. 作为状态变量,樀仅在平衡时定义。平衡状态之间的樀变化 $(A, B)$ 通过 积分其微分,方程找到。(1.8),
$$
S(B)-S(A)=\int_{A}^{B}(\mathrm{~d} Q)_{\mathrm{rev}} / T
$$
对于任何连接的路径 $A$ 和 $B$ ,使得热传递在所有阶段都可逆地发生。 ${ }^{24}$
统计代写请认准statistics-lab™. statistics-lab™为您的留学生涯保驾护航。
金融工程代写
金融工程是使用数学技术来解决金融问题。金融工程使用计算机科学、统计学、经济学和应用数学领域的工具和知识来解决当前的金融问题,以及设计新的和创新的金融产品。
非参数统计代写
非参数统计指的是一种统计方法,其中不假设数据来自于由少数参数决定的规定模型;这种模型的例子包括正态分布模型和线性回归模型。
广义线性模型代考
广义线性模型(GLM)归属统计学领域,是一种应用灵活的线性回归模型。该模型允许因变量的偏差分布有除了正态分布之外的其它分布。
术语 广义线性模型(GLM)通常是指给定连续和/或分类预测因素的连续响应变量的常规线性回归模型。它包括多元线性回归,以及方差分析和方差分析(仅含固定效应)。
有限元方法代写
有限元方法(FEM)是一种流行的方法,用于数值解决工程和数学建模中出现的微分方程。典型的问题领域包括结构分析、传热、流体流动、质量运输和电磁势等传统领域。
有限元是一种通用的数值方法,用于解决两个或三个空间变量的偏微分方程(即一些边界值问题)。为了解决一个问题,有限元将一个大系统细分为更小、更简单的部分,称为有限元。这是通过在空间维度上的特定空间离散化来实现的,它是通过构建对象的网格来实现的:用于求解的数值域,它有有限数量的点。边界值问题的有限元方法表述最终导致一个代数方程组。该方法在域上对未知函数进行逼近。[1] 然后将模拟这些有限元的简单方程组合成一个更大的方程系统,以模拟整个问题。然后,有限元通过变化微积分使相关的误差函数最小化来逼近一个解决方案。
tatistics-lab作为专业的留学生服务机构,多年来已为美国、英国、加拿大、澳洲等留学热门地的学生提供专业的学术服务,包括但不限于Essay代写,Assignment代写,Dissertation代写,Report代写,小组作业代写,Proposal代写,Paper代写,Presentation代写,计算机作业代写,论文修改和润色,网课代做,exam代考等等。写作范围涵盖高中,本科,研究生等海外留学全阶段,辐射金融,经济学,会计学,审计学,管理学等全球99%专业科目。写作团队既有专业英语母语作者,也有海外名校硕博留学生,每位写作老师都拥有过硬的语言能力,专业的学科背景和学术写作经验。我们承诺100%原创,100%专业,100%准时,100%满意。
随机分析代写
随机微积分是数学的一个分支,对随机过程进行操作。它允许为随机过程的积分定义一个关于随机过程的一致的积分理论。这个领域是由日本数学家伊藤清在第二次世界大战期间创建并开始的。
时间序列分析代写
随机过程,是依赖于参数的一组随机变量的全体,参数通常是时间。 随机变量是随机现象的数量表现,其时间序列是一组按照时间发生先后顺序进行排列的数据点序列。通常一组时间序列的时间间隔为一恒定值(如1秒,5分钟,12小时,7天,1年),因此时间序列可以作为离散时间数据进行分析处理。研究时间序列数据的意义在于现实中,往往需要研究某个事物其随时间发展变化的规律。这就需要通过研究该事物过去发展的历史记录,以得到其自身发展的规律。
回归分析代写
多元回归分析渐进(Multiple Regression Analysis Asymptotics)属于计量经济学领域,主要是一种数学上的统计分析方法,可以分析复杂情况下各影响因素的数学关系,在自然科学、社会和经济学等多个领域内应用广泛。
MATLAB代写
MATLAB 是一种用于技术计算的高性能语言。它将计算、可视化和编程集成在一个易于使用的环境中,其中问题和解决方案以熟悉的数学符号表示。典型用途包括:数学和计算算法开发建模、仿真和原型制作数据分析、探索和可视化科学和工程图形应用程序开发,包括图形用户界面构建MATLAB 是一个交互式系统,其基本数据元素是一个不需要维度的数组。这使您可以解决许多技术计算问题,尤其是那些具有矩阵和向量公式的问题,而只需用 C 或 Fortran 等标量非交互式语言编写程序所需的时间的一小部分。MATLAB 名称代表矩阵实验室。MATLAB 最初的编写目的是提供对由 LINPACK 和 EISPACK 项目开发的矩阵软件的轻松访问,这两个项目共同代表了矩阵计算软件的最新技术。MATLAB 经过多年的发展,得到了许多用户的投入。在大学环境中,它是数学、工程和科学入门和高级课程的标准教学工具。在工业领域,MATLAB 是高效研究、开发和分析的首选工具。MATLAB 具有一系列称为工具箱的特定于应用程序的解决方案。对于大多数 MATLAB 用户来说非常重要,工具箱允许您学习和应用专业技术。工具箱是 MATLAB 函数(M 文件)的综合集合,可扩展 MATLAB 环境以解决特定类别的问题。可用工具箱的领域包括信号处理、控制系统、神经网络、模糊逻辑、小波、仿真等。
